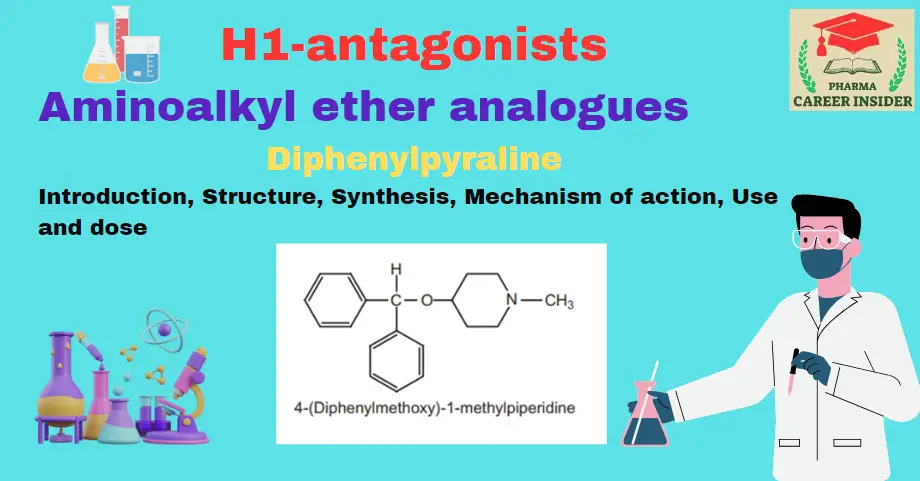
Introduction
Pharmaceutical analysis is a branch of analytical chemistry that focuses on the identification, determination, quantification, and quality control of substances used in the production of pharmaceuticals, as well as the analysis of pharmaceutical products themselves. Its primary aim is to ensure the safety, efficacy, and quality of drugs and pharmaceutical preparations. Here’s a note on the definition and scope of pharmaceutical analysis:
Definition:
Pharmaceutical analysis is a branch of pharmacy and analytical chemistry that focuses on the identification, quantification, and characterization of drugs, their excipients, and related substances. It involves various qualitative and quantitative analytical techniques to ensure the quality, purity, potency, efficacy, and safety of pharmaceutical products.
or
Pharmaceutical analysis is the systematic use of analytical techniques on pharmaceutical substances, drugs, and products. It involves evaluating the identity, purity, quality, and stability of pharmaceutical materials and products during their development, manufacturing, and distribution.
Scope of Pharmaceutical Analysis
Pharmaceutical analysis plays a crucial role in different stages of drug development, formulation, quality control, and regulatory compliance. Its scope extends across various areas, including:
1. Qualitative and Quantitative Analysis
- Qualitative Analysis: Determines the identity of a compound or drug using techniques such as spectroscopy, chromatography, and chemical tests.
- Quantitative Analysis: Measures the concentration or amount of a drug in a given sample using titration, UV-visible spectrophotometry, high-performance liquid chromatography (HPLC), and other advanced techniques.
2. Drug Discovery and Development
- Helps in the identification and characterization of new drug molecules.
- Supports the optimization of drug formulations.
- Ensures the stability of drug compounds during development.
3. Quality Control (QC) and Quality Assurance (QA)
- Ensures that pharmaceutical products meet the required standards of purity, potency, and safety.
- Helps in batch-to-batch consistency of drug formulations.
- Follows guidelines set by regulatory agencies like USFDA, WHO, ICH, and Pharmacopeias (USP, BP, IP).
4. Regulatory and Pharmacopoeial Analysis
- Compliance with official pharmacopeial standards such as USP (United States Pharmacopeia), BP (British Pharmacopeia), and IP (Indian Pharmacopeia).
- Ensures that pharmaceutical products meet international regulatory requirements before market approval.
5. Stability Testing
- Evaluates the stability of drugs under various environmental conditions such as temperature, humidity, and light exposure.
- Helps in determining the shelf life and storage conditions of pharmaceutical products.
6. Impurity Profiling and Contaminant Detection
- Identifies and quantifies impurities, degradation products, and residual solvents in pharmaceutical substances.
- Ensures drug safety by detecting toxic contaminants.
7. Bioanalytical Applications
- Used in pharmacokinetic and pharmacodynamic studies to measure drug levels in biological fluids (plasma, urine, blood).
- Supports clinical trials by ensuring drug efficacy and safety.
8. Drug Formulation and Development
- Assists in the selection of appropriate excipients and formulation strategies.
- Ensures the uniformity of active pharmaceutical ingredients (APIs) in dosage forms.
9. Analytical Method Validation
- Confirms that an analytical method is suitable, accurate, precise, and reproducible for its intended purpose.
- Follows guidelines set by ICH (International Council for Harmonisation) and regulatory bodies.
10. Advanced Analytical Techniques
Pharmaceutical analysis employs several modern techniques for accurate and sensitive analysis:
- Spectroscopic Methods: UV-visible spectroscopy, Infrared Spectroscopy (IR), Nuclear Magnetic Resonance (NMR), Mass Spectrometry (MS).
- Chromatographic Methods: High-Performance Liquid Chromatography (HPLC), Gas Chromatography (GC), Thin-Layer Chromatography (TLC).
- Electrochemical Methods: Potentiometry, Conductometry, Voltammetry.
Conclusion
Pharmaceutical analysis is an essential aspect of drug discovery, development, and production. It ensures the safety, efficacy, and quality of pharmaceutical products through rigorous testing and validation methods. By integrating modern analytical techniques, pharmaceutical analysis continues to evolve, meeting the increasing demands of global regulatory standards and patient safety.